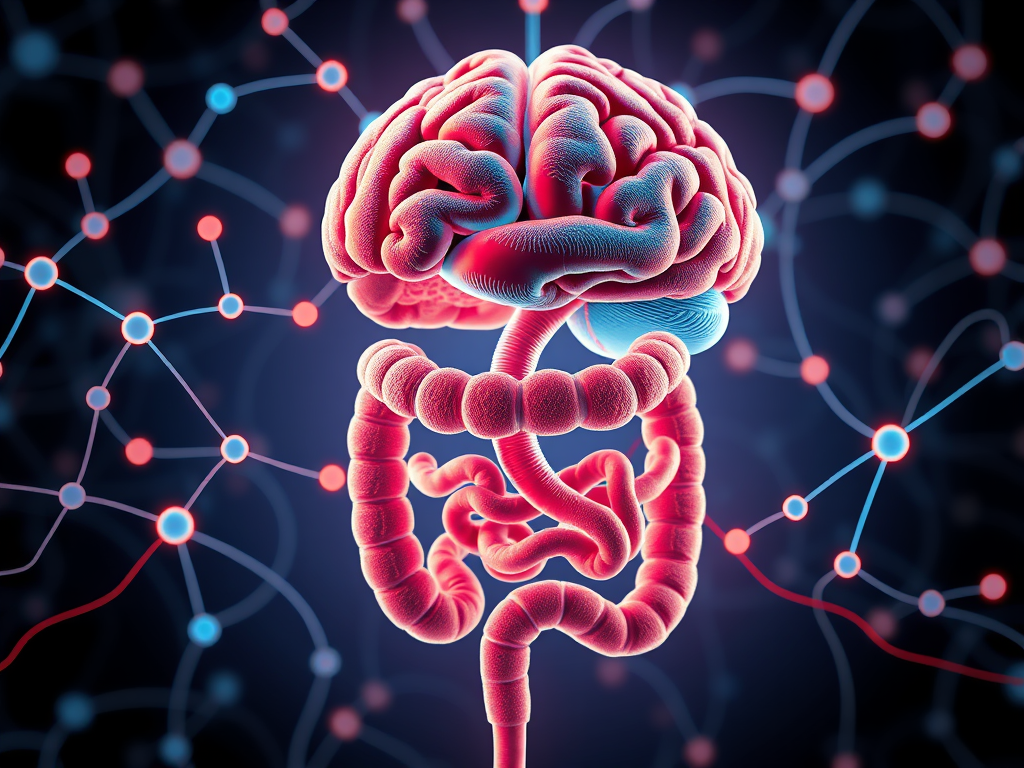
Do you know our body constantly sends signals to our brain that affect our moods? One of the most intriguing findings that scientists discovered was that micro-bacterial flora in the guts can affect communication between our intestines and brain leading to mood disorders via the gut-brain axis.
The gut-brain axis (GBA) refers to the bidirectional communication system between gut microbiota, or microbiome, and the brain. The gut refers to the gastrointestinal tract (GI), which includes organs such as the stomach and small and large intestines, and the gut microbiota refers to the trillions of bacteria that are present within the gut. This communication system uses three main pathways: neural (through neurons), inflammatory (through cytokines and immune cells), and humoral (through the HPA axis, consisting of the Hypothalamus, Pituitary glands, and Adrenal glands). The gut-brain axis has been shown to be interrelated with a variety of mental disorders, including major depressive disorder (MDD), autism, and anxiety disorders.
It has been suggested that the inflammation that is seen in depressed patients is a result of changes at the microbiota levels or at least correlated. There is evidence of microbiome alterations in depressed patients, which suggests that they may contribute to dysregulated inflammatory responses. Increased signaling in the Nucleotide-binding domain, Leucine-rich–containing family, Pyrin domain–containing-3, or alternatively NLR family Pyrin domain containing 3 (NLRP3) inflammasome, a protein involved in the immune response, led to an increase in pro-inflammatory markers within gut-microbiota. Another study showed that fecal microbiota transplantation in animals with depression induced by chronic unpredictable stress (CUS) was able to restore decreased levels of serotonin and suppress activity of microglia and astrocytes as well as reduce expression of NLRP3 and other pro-inflammatory cytokines within the hippocampus of the animals.
Apart from inflammation, the gut microbiome has been implicated in depression through its ability to increase Brain-Derived Neurotrophic Factor (BDNF) expression in the brain. BDNF increases neurogenesis, which is the production of new neurons, and has been shown to be strongly decreased in depression. In one study, the addition of two strains of bacteria into the gut microbiota was able to increase BDNF levels in patients with MDD and other psychiatric disorders. Overall, the gut-brain axis holds potential as a pathway to mediating inflammation and depression.
The gut-brain axis and microbiome is linked to inflammation. Furthermore, increased NLRP3 signaling is commonly seen in patients with MDD, and this increases the amount of inflammatory markers within gut-microbiota. Fecal microbiota transplantation suppressed the activity of microglia and astrocytes and reduced expression of NLRP3 and pro-inflammatory cytokine IL-1b in the HPC of chronic unpredictable stress (CUS)-exposed animals, indicating that this axis plays a significant role in depression.
The antidepressant effects of ketamine may be mediated by its alteration of gut microbiota composition. Butyrate is a short-chain fatty acid–produced by bacterial fermentation in the gut–that can reduce inflammation. Ketamine administration increases butyrate producing microbiota including Butyricimonas, Clostridiales, Turicibacter and Bacillus. It also reduces several kinds of bacteria that contribute to inflammation.
Source: Roger Li Curieux Academic Journal, issue 50